
There are few industries where the importance of product conformity is as crucial as the design, manufacture, and testing of medical devices.
Manufacturers of any medical device systems must meet specific requirements for the safety and effectiveness of the medical products they sell.
The certification of a quality management system, specific for Medical Devices will be to your advantage. In many cases, it is a requirement for customers or organizations who manufacture medium and higher class medical devices and export their products to the global market such as Europe and Canada.
ISO 13485 is a stand-alone standard. The ISO 13485:2016 is based on ISO 9001:2008 or ISO 9000. It is intended to promote global alignment of appropriate regulatory requirements specific to quality/medical device quality systems for organizations.
Such organizations can be involved in one or more stages of the life-cycle. They include design and development, production, storage and distribution, installation, servicing of a medical device, and design and development or provision of associated activities (e.g. technical support). These requirements are critical for the medical device industry.
Or are your customers requiring this additional compliance audit/certification? We can help.
Our auditors have extensive hands on experience in the Medical Device industry. You will be working with ISO audit professionals.
We currently do not do 3rd party registration to ISO 13485. We do perform value added compliance audits to medical using lead auditors with knowledge and experience in ISO 13485.
Don’t let compliance and quality concerns hold back your medical device manufacturing business. Contact DAC Audit Services today to learn more about our medical device internal audits, medical device systems, and medical device registration services. We’re here to be your trusted partner in achieving compliance and quality in the medical device industry.
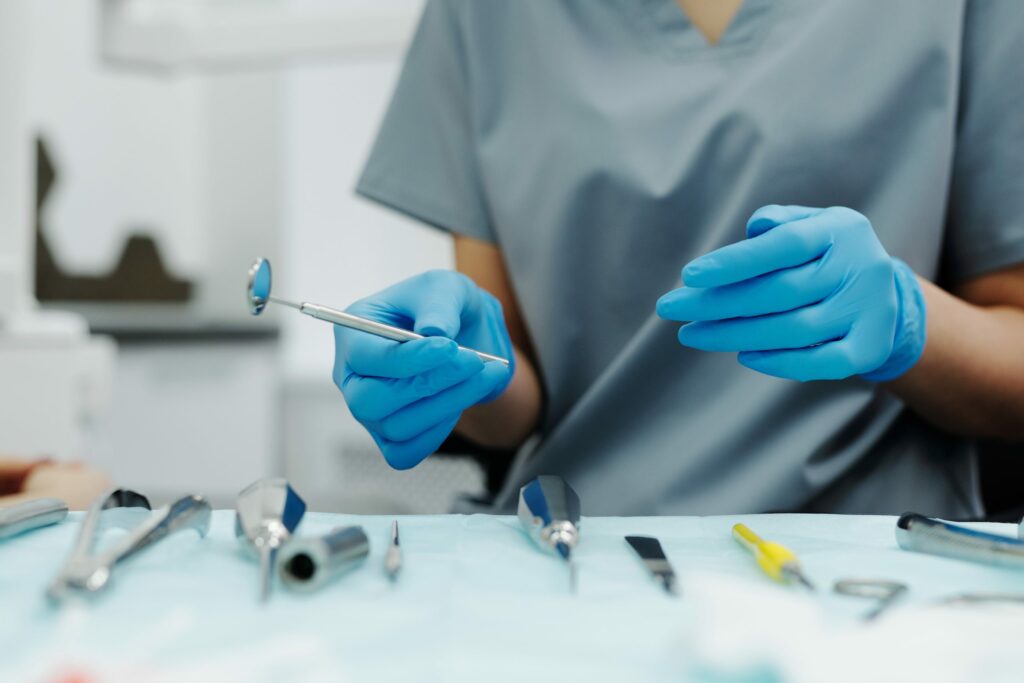

Are you concerned about the quality and compliance of your medical device manufacturing processes? Do you worry about potential regulatory violations or inefficiencies in your operations? These concerns can keep you up at night and keep your business from achieving its full potential. At DAC Audit Services, we understand the pain points of medical device manufacturing, so we offer medical device internal audit services. Our experienced auditors can conduct thorough audits of your internal processes, identifying gaps in your compliance and providing recommendations for improvement. With our help, you can achieve optimal performance and quality in your medical device manufacturing operations, giving you peace of mind and ensuring your business’s success. Don’t let compliance concerns hold back your medical device manufacturing business.
Do you struggle with ensuring compliance and quality in your medical device manufacturing operations? Are you unsure about the medical device systems and processes required to meet regulatory and industry standards? These concerns can hold back your business and put you at risk of regulatory violations and product defects. At DAC Audit Services, we understand the pain points that come with medical device systems, which is why we offer guidance and support to help you implement the necessary systems for your operations. Our experienced team can help you with design control, risk management, quality management systems, and more. With our help, you can achieve compliance and quality in your medical device manufacturing operations, ensuring your business’s success and reputation. Don’t let compliance and quality concerns hold back your medical device manufacturing business.
Are you struggling to navigate the complex registration process for your medical device products? Do you need guidance and support to meet all regulatory requirements for your specific market? These concerns can be overwhelming and put your business at risk of delays or rejections in the registration process. At DAC Audit Services, we understand the pain points that come with medical device registration, which is why we offer a range of registration services to help you achieve compliance and gain market access. Our team of experts has a thorough understanding of the regulatory requirements for medical devices and can guide you through the registration process, ensuring that you have the necessary documentation and certifications for your specific market. With our help, you can achieve regulatory compliance, gain market access, and avoid delays or rejections in the registration process. Don’t let registration concerns hold back your medical device systems manufacturing business. Contact DAC Audit Services today.
Are you looking for a trusted partner to help you achieve compliance and quality in your medical device manufacturing operations? Do you need expert guidance and support to navigate the complex regulatory landscape for medical devices? These concerns can be daunting and require a partner with the expertise and experience to help you succeed. At DAC Audit Services, we understand the pain points that come with medical device compliance and quality, which is why we offer a range of services to help you achieve your goals. Our team of experts has a thorough understanding of the regulatory landscape for medical devices and can provide the guidance and support you need to achieve compliance and quality in your operations. We are committed to being a trusted partner to our clients and delivering exceptional service and support throughout the process. With our help, you can succeed in the medical device industry and build a reputation for quality and compliance. Don’t settle for anything less than a trusted partner for your medical device compliance and quality needs.

Our primary focus is to standards such as ISO 9001, ISO 14001, ISO 13485, ISO 18001, and more. We also audit to other standards as well, depending on your company’s or vendor’s needs.
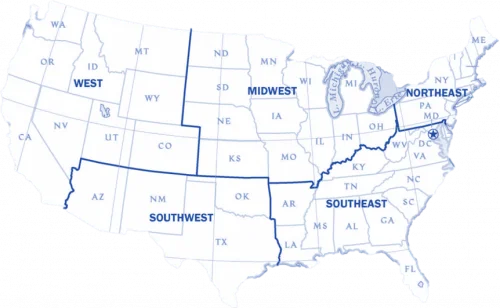
We serve the East, Midwest, West, South and Southwest regions of the United States. Please visit our Contact Us page for more information or call us at 1-978-386-2516.
1-978-386-2516
Info@DACAudit.com
22 County Road, Suite #101 Ashby, MA 01431